Ertan, Salih
Loading...
Profile URL
Name Variants
Ertan, Salih
Ertan,Salih
S., Ertan
Ertan,S.
E.,Salih
Salih Ertan
Salih, Ertan
E., Salih
S.,Ertan
Ertan,Salih
S., Ertan
Ertan,S.
E.,Salih
Salih Ertan
Salih, Ertan
E., Salih
S.,Ertan
Job Title
Doktor Öğretim Üyesi
Email Address
salih.ertan@tilim.edu.tr
Main Affiliation
Chemical Engineering
Status
Website
ORCID ID
Scopus Author ID
Turkish CoHE Profile ID
Google Scholar ID
WoS Researcher ID
Sustainable Development Goals

Documents
9
Citations
162
h-index
7

Documents
10
Citations
169

Scholarly Output
10
Articles
9
Views / Downloads
68/142
Supervised MSc Theses
1
Supervised PhD Theses
0
WoS Citation Count
121
Scopus Citation Count
124
Patents
0
Projects
0
WoS Citations per Publication
12.10
Scopus Citations per Publication
12.40
Open Access Source
0
Supervised Theses
1
| Journal | Count |
|---|---|
| Dyes and Pigments | 3 |
| Electrochimica Acta | 2 |
| Journal of Polymer Science Part A: Polymer Chemistry | 1 |
| Macromolecules | 1 |
| Materials Today Communications | 1 |
Current Page: 1 / 2
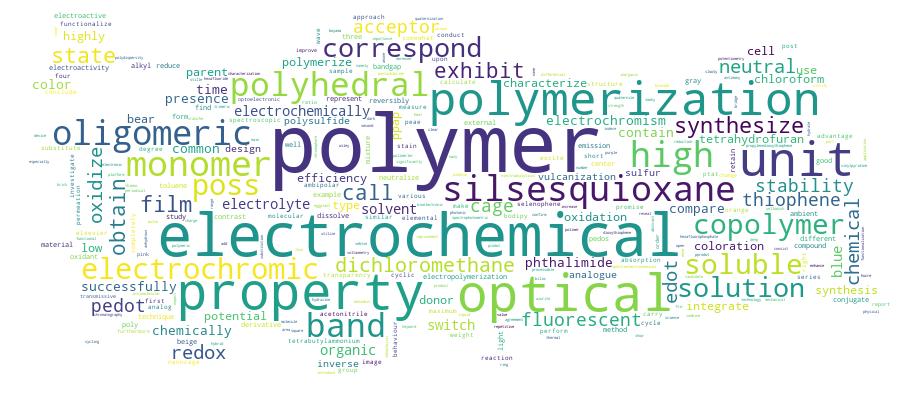
Competency Cloud